Snowflakes
Page 1 of 1
 Snowflakes
Snowflakes
Snowflakes
Main article: Snowflake
Snowflake viewed in an optical microscope
Snow crystals form when tiny supercooled cloud droplets (about 10 μm in diameter) freeze. These droplets are able to remain liquid at temperatures lower than −18 °C (0 °F), because to freeze, a few molecules in the droplet need to get together by chance to form an arrangement similar to that in an ice lattice; then the droplet freezes around this "nucleus." Experiments show that this "homogeneous" nucleation of cloud droplets only occurs at temperatures lower than −35 °C (−31 °F).[21] In warmer clouds an aerosol particle or "ice nucleus" must be present in (or in contact with) the droplet to act as a nucleus. Our understanding of what particles make efficient ice nuclei is poor what we do know is they are very rare compared to that cloud condensation nuclei on which liquid droplets form. Clays, desert dust and biological particles may be effective,[22] although to what extent is unclear. Artificial nuclei include particles of silver iodide and dry ice, and these are used to stimulate precipitation in cloud seeding.[23]
Once a droplet has frozen, it grows in the water-supersaturated air, when the temperature remains below the freezing point. The droplet then grows by condensation of water vapor onto the ice surfaces. Air saturation with water is maintained by continuous simultaneous evaporation of water droplets. Thus ice crystals grow at the expense of water droplets in a process called the Wegner-Bergeron-Findeison process. These large crystals are an efficient source of precipitation, since they fall through the atmosphere due to their weight, and may collide and aggregate in clusters. These aggregates are snowflakes, and are usually the type of ice particle that falls to the ground.[24] Guinness World Records list the worlds largest snowflakes as those of January 1887 at Fort Keogh, Montana; allegedly one measured 38 cm (15 inches) wide.[25]
The exact details of the sticking mechanism remain controversial. Possibilities include mechanical interlocking, sintering, electrostatic attraction as well as the existence of a "sticky" liquid-like layer on the crystal surface. The individual ice crystals often have hexagonal symmetry. Although the ice is clear, scattering of light by the crystal facets and hollows/imperfections mean that the crystals often appear white in colour due to diffuse reflection of the whole spectrum of light by the small ice particles.[26] The shape of the snowflake is determined broadly by the temperature and humidity at which it is formed.[24] Rarely, at a temperature of around −2 °C (28 °F), snowflakes can form in threefold symmetry triangular snowflakes.[27] The most common snow particles are visibly irregular, although near-perfect snowflakes may be more common in pictures because they are more visually appealing. It is unlikely that any two snowflakes are alike due to the estimated 10,000,000,000,000,000,000 water molecules which make up a typical snowflake,[28] which grow at different rates and in different patterns depending on the changing temperature and humidity within the atmosphere that the snowflake falls through on its way to the ground.[29] The METAR code for snow is SN, while snow showers are coded SHSN.[10]
Divorce Lawyer New York
positive cashflow property
Main article: Snowflake
Snowflake viewed in an optical microscope
Snow crystals form when tiny supercooled cloud droplets (about 10 μm in diameter) freeze. These droplets are able to remain liquid at temperatures lower than −18 °C (0 °F), because to freeze, a few molecules in the droplet need to get together by chance to form an arrangement similar to that in an ice lattice; then the droplet freezes around this "nucleus." Experiments show that this "homogeneous" nucleation of cloud droplets only occurs at temperatures lower than −35 °C (−31 °F).[21] In warmer clouds an aerosol particle or "ice nucleus" must be present in (or in contact with) the droplet to act as a nucleus. Our understanding of what particles make efficient ice nuclei is poor what we do know is they are very rare compared to that cloud condensation nuclei on which liquid droplets form. Clays, desert dust and biological particles may be effective,[22] although to what extent is unclear. Artificial nuclei include particles of silver iodide and dry ice, and these are used to stimulate precipitation in cloud seeding.[23]
Once a droplet has frozen, it grows in the water-supersaturated air, when the temperature remains below the freezing point. The droplet then grows by condensation of water vapor onto the ice surfaces. Air saturation with water is maintained by continuous simultaneous evaporation of water droplets. Thus ice crystals grow at the expense of water droplets in a process called the Wegner-Bergeron-Findeison process. These large crystals are an efficient source of precipitation, since they fall through the atmosphere due to their weight, and may collide and aggregate in clusters. These aggregates are snowflakes, and are usually the type of ice particle that falls to the ground.[24] Guinness World Records list the worlds largest snowflakes as those of January 1887 at Fort Keogh, Montana; allegedly one measured 38 cm (15 inches) wide.[25]
The exact details of the sticking mechanism remain controversial. Possibilities include mechanical interlocking, sintering, electrostatic attraction as well as the existence of a "sticky" liquid-like layer on the crystal surface. The individual ice crystals often have hexagonal symmetry. Although the ice is clear, scattering of light by the crystal facets and hollows/imperfections mean that the crystals often appear white in colour due to diffuse reflection of the whole spectrum of light by the small ice particles.[26] The shape of the snowflake is determined broadly by the temperature and humidity at which it is formed.[24] Rarely, at a temperature of around −2 °C (28 °F), snowflakes can form in threefold symmetry triangular snowflakes.[27] The most common snow particles are visibly irregular, although near-perfect snowflakes may be more common in pictures because they are more visually appealing. It is unlikely that any two snowflakes are alike due to the estimated 10,000,000,000,000,000,000 water molecules which make up a typical snowflake,[28] which grow at different rates and in different patterns depending on the changing temperature and humidity within the atmosphere that the snowflake falls through on its way to the ground.[29] The METAR code for snow is SN, while snow showers are coded SHSN.[10]
Divorce Lawyer New York
positive cashflow property
meodingu- Gender :
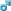
Locale : hp vn
Kapisanan : Binhi
Page 1 of 1
Permissions in this forum:
You cannot reply to topics in this forum